Advancing an innovative technology
When a person has to endure symptoms of mental illness because current treatments don’t work for them, the need for alternatives is obvious.
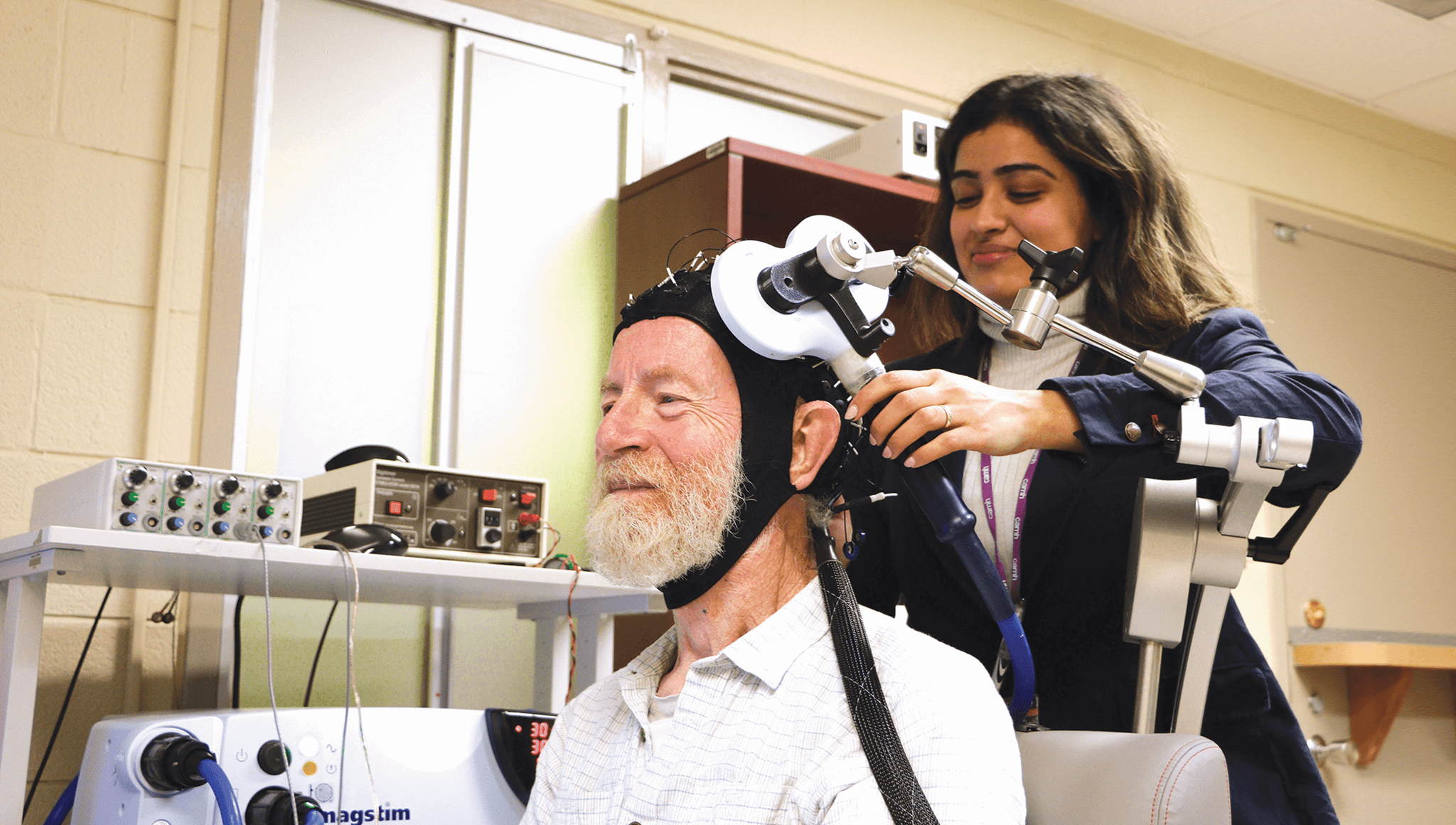
CAMH has been a pioneer in the use of brain stimulation, one of the few new treatment options for mental illness in the past 30 years. CAMH’s Temerty Centre for Therapeutic Brain Intervention is leading innovative studies that suggest this therapeutic approach, which targets the brain’s neural circuits, holds promise for many mental illnesses.
For hard-to-treat depression, repetitive transcranial magnetic stimulation (rTMS) is known to be effective, but the course of treatment, at 45 minutes daily for up to six weeks, is time-consuming and cost-intensive. In a major advance, a study led by Dr. Daniel Blumberger has shown that a new “theta-burst” form of rTMS that takes only three minutes is just as effective as the standard.
Different forms of brain stimulation, including rTMS, transcranial direct current stimulation (tDCS) and magnetic seizure therapy (MST), are being studied as both treatment and investigative tools. “We’re aiming to identify and study biological targets for treatment, and to understand the brain mechanisms underlying disorders,” says Dr. Jeff Daskalakis.
While brain stimulation is not yet a household term, the range of promising studies underway at CAMH suggests it will be in the future. Researchers are exploring the use of brain stimulation to improve memory in schizophrenia, reduce cravings in cannabis addiction, prevent Alzheimer’s dementia, treat post-traumatic stress disorder and mild autism, and more.